This loss of chromium may inhibit gelation and limit the depth of penetration of a gel system in dolomite matrix rock. The experimental results indicate that chromium is precipitated at a relatively high rate when chromium acetate solution is in contact with dolomite core material. Precipitation occurred faster in the cores than in the batch experiments. However, the induction period observed in bulk solutions was not present in the data taken in the cores. The kinetic model fit the precipitation rate data well and is consistent with the conclusion that precipitation is the principal retention mechanism. The retention capacity of the rock appeared to be infinite.Ĭhromium concentrations at various residence times were compared to values predicted from a kinetic model developed for precipitation from bulk solutions. At the lowest flow rate used, effluent chromium concentration approached a steady-state value that was much smaller than the injected concentration. In the range of concentrations studied, 0 to 1% by weight, the presence of KCl increased the retention rate. In the continuous displacements, effluent concentration profiles were a function of flow rate, indicating that the retention was a rate-dependent process. There was significant retention of chromium in the core in both continuous and shut-in experiments.

In some experiments, the cores were shut in and the resident fluid displaced and analyzed to determine chromium retention during the shut-in period. Displacement experiments were conducted in which chromium(III) acetate solutions were continuously displaced at different flow rates through Baker dolomite cores with permeabilities from 19 to 25 md. This paper describes a study of chromium retention in dolomite cores in the absence of polymer and demonstrates that precipitation of chromium is the principal retention mechanism. In-depth treatment of the carbonate rock matrix by these systems is of concern because rock-fluid interactions can lead to loss of chromium and may limit the penetration of a gel treatment. Also, preferably, after production of the chromium (III) carbonate, filtration is performed, and the chromium (III) carbonate is washed with water until the conductivity of the filtrate is 5 mS/cm or less.Chromium(III)-polymer gel systems are often used in permeability modification treatments for flow control. 4H 2 O precipitates the silver chloride when treated with silver nitrate.
Chromium iii carbonate formula skin#
Despite trivalent chromium being far less poisonous than hexavalent, it should be handled carefully as it can cause an allergic skin reaction to a living being.
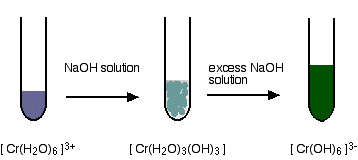
This chromium (III) carbonate is preferably obtained by contacting an aqueous solution of carbonate and an aqueous solution containing trivalent chromium at a pH of 6 to 12 under the condition of a reaction liquid temperature of 0° C. The Chromium (III) Chloride formula is CrCl 3. This chromium (III) carbonate is preferably completely dissolved within 30 minutes when the chromium (III) carbonate is added, in an amount corresponding to a Cr content of 1 g, to 1 liter of an aqueous solution of hydrochloric acid having a pH of 0.2 at a temperature of 25° C. This chromium (III) carbonate has an L* value of 50 to 70, an a* value of ?4 to ?2, and a b* value of ?10 to ?7, which are represented by the L*a*b* color system (JIS Z8729). Abstract: The chromium (III) carbonate of the present invention exhibits a light blue color in a solid state.
